ELISA Kits
- • Anemia ELISA kits
- • Allergy ELISA kits
- • Autoimmune Disease kits
- • Bone Metabolism ELISA kits
- • Blood bank ELISA kits
- • Cancer ELISA kits
- • Cardiac Markers ELISA kits
- • Diabetes Assays ELISA kits
- • Drug test ELISA kits
- • Fertility ELISA kits
- • Food ELISA kits
- • Infectious Disease ELISA kits
- • Other ELISA Kits
- • Parasitology ELISA kits
- • Steroid ELISA kits
- • Thyroid ELISA kits
Rapid Tests
- • Allergy Rapid tests
- • Bone Metabolism
- • Cancer Rapid tests
- • Cardiac markers Rapid tests
- • Drug Tests
- • Fertility Rapid tests
- • Hepatitis Panel
- • Infectious Disease & other tests
- • Other
- • Ovulation Rapid tests
- • Pregnancy tests
- • Urine Reagent Strips tests
IFA Kits
Chemiluminescence Immuno Assays
- • Allergy Assays
- • Autoimmune Thyroid Assays
- • Cardio-Vascular Monitoring
- • Diabetes Assays
- • Fertility Assays
- • Growth Deficiency
- • Infectious Disease Assays
- • Others
- • Steroid Assays
- • Thyroid Assays
- • Tumor Marker Assays
Serology Tests
- • ASO (Anti-Streptolysin-O)
- • CRP (C-Reactive Protein)
- • Mono (Infectious Mononucleosis)
- • RF (Rheumatoid Factor)
- • RPR (Rapid Plasma Reagin)
- • SLE (Systemic Lupus Erythematosus)
Instrumentation



Chlamydia Rapid Test (Female)
| Name |
Chlamydia Rapid Test (Cassette) |
|---|---|
| Full name |
Human Chlamydia RapiCard Insta Test (Cassette) Female, Export Use Only |
| Category Name | Ovulation Rapid tests |
| Test | Boxed in 20 tests |
| Specificity | 99.9% |
| Sensitivity | 94.6% |
 |
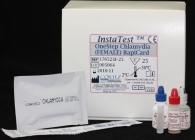 |
 |
Chlamydia Rapid Test (Female) description:
"Export Use Only"
Diagnostic Automation offers a OneStep Chlamydia RapiCard InstaTest is a rapid immunoassay for direct qualitative detection of Chlamydia trachomatis antigen endocervical or endourethral swab specimens. The Chlamydia Rapid Test is a meaningful test for point-of-care diagnosis of a chlamydia infection. This Chlamydia Rapid Test is a quick, simple, and non-invasive test for use on female patients only.
Materials Provided with Chlamydia Rapid Test kit:
1. Testing Devices
2. Extraction Buffer A
3. Extraction Buffer B
4. Positive Control
5. Extraction Tube
6. Test Instructions
7. Female Swab
Materials required, not provided:
1. Timer
2. Test tube rack
Chlamydia Rapid Test Background Information
Chlamydia trachomatis is one of the most common sexually transmitted pathogens. It is a major
cause of cervicitis, urethritis, endometritis, and pelvic inflammatory disease in women. Serious complications can result in salpingitis, infertility, and ectopic pregnancy. If transmitted to infants
during birth, Chlamydia can cause conjunctivitis and pneumonia. 50-70% of infected women are asymptomatic, which makes diagnosis extremely important. The primary method for detection of Chlamydia is growth of the organism in cell culture. Other methods include direct fluorescence assays (DFA), enzyme immunoassays (EIA), and nucleic acid probing.
Chlamydia Rapid Test Principle
The OneStep Chlamydia Rapid Test utilizes the chemical extraction of a carbohydrate antigen from Chlamydia followed by the utilization of migratory color immunoassay technology for the
qualitative detection of Chlamydia trachomatis. In the test procedure, a unique set of polyclonal and monoclonal antibodies are employed. One antibody is immobilized on a porous membrane
while the other antibody is conjugated to dye particles as signaling component. If using swab, specimen or from a patient is treated with extraction reagent A and B to extract the antigen.
The test dipstick is then immersed in the extraction sample. The liquid migrates through the absorbent area and along the membrane. If Chlamydia trachomatis antigen is present, the labeled antibody dye conjugate bind to it, forming an antibody antigen complex. For additional details please refer to the instructions for use.
Notable features of Chlamydia Rapid Test:
reliable and inexpensive
easy-to-understand directions
clear reagent and specimen preparation procedures
simple test kit storage instructions
safe and convenient rapid test kit packaging
rapid and accurate test results - within 5 minutes
See Chlamydia Rapid Test Product Insert for specific details on preparation, procedures, quality control, and interpretation of test results.
Diagnostic Automation Inc. also offers other Infectious Diseases Rapid tests, and Infectious Diseases ELISA test kits. For more information about Rapid tests, CLIA tests, IFA kits, or ELISA test kits, please contact our Customer Service Representatives at 818-591-3030 .
*Not to be sold, shipped, or distributed in the United States.
ELISA kits - Rapid tests- Drug tests- Pregnancy test - IFA kits - CLIA assays - Serology tests - Instrumentation
©1992 Diagnostic Automation/Cortez Diagnostics Inc. All rights reserved.