ELISA Kits
- • Anemia ELISA kits
- • Allergy ELISA kits
- • Autoimmune Disease kits
- • Bone Metabolism ELISA kits
- • Blood bank ELISA kits
- • Cancer ELISA kits
- • Cardiac Markers ELISA kits
- • Diabetes Assays ELISA kits
- • Drug test ELISA kits
- • Fertility ELISA kits
- • Food ELISA kits
- • Infectious Disease ELISA kits
- • Other ELISA Kits
- • Parasitology ELISA kits
- • Steroid ELISA kits
- • Thyroid ELISA kits
Rapid Tests
- • Allergy Rapid tests
- • Bone Metabolism
- • Cancer Rapid tests
- • Cardiac markers Rapid tests
- • Drug Tests
- • Fertility Rapid tests
- • Hepatitis Panel
- • Infectious Disease & other tests
- • Other
- • Ovulation Rapid tests
- • Pregnancy tests
- • Urine Reagent Strips tests
IFA Kits
Chemiluminescence Immuno Assays
- • Allergy Assays
- • Autoimmune Thyroid Assays
- • Cardio-Vascular Monitoring
- • Diabetes Assays
- • Fertility Assays
- • Growth Deficiency
- • Infectious Disease Assays
- • Others
- • Steroid Assays
- • Thyroid Assays
- • Tumor Marker Assays
Serology Tests
- • ASO (Anti-Streptolysin-O)
- • CRP (C-Reactive Protein)
- • Mono (Infectious Mononucleosis)
- • RF (Rheumatoid Factor)
- • RPR (Rapid Plasma Reagin)
- • SLE (Systemic Lupus Erythematosus)
Instrumentation



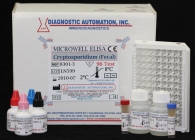
Cryptosporidium Fecal ELISA kit
| Name |
Cryptosporidium Fecal ELISA Test |
|---|---|
| Full name |
Human Cryptosporidium Fecal ELISA Test Kit |
| Category Name | Diabetes Assays ELISA kits |
| Test | 96 |
| Method | ELISA method: Enzyme Linked Immunosorbent Assay |
| Principle | ELISA principle- Sandwhich; Antibody Coated Plate |
| Detection Range | Qualitative - Positive, Negative Controls |
| Sample | 1gm Stool Sample |
| Specificity | 100% |
| Sensitivity | 100% |
| Total Time | ~100min |
| Shelf Life | 18 months from the manufacturing date |
 |
 |
 |
Cryptosporidium Fecal ELISA kit description:
Cryptosporidium ELISA Kit is an in vitro immunoassay for the qualitative determination of Cryptosporidium antigen in feces.
Material Provided with Chagas ELISA kit:
1. Test Strips Microwells: 96 test wells anti-Cryptosporidium polyclonal antibodies
2. Enzyme Conjugate: anti-Cryptosporidium antibodies conjugated-HRP
3. Positive Control: diluted Cryptosporidium positive formalinized stool supernatant
4. Negative Control: diluted Cryptosporidium negative formalinized stool supernatant
5. TMB Substrate Solution: TMB and peroxid
6. Wash Concentrate 20X
7. Dilution Buffer: buffered protein solution containing Thimerosal
8. Stop Solution: 5% phosphoric acid
Materials Required, Not Provided:
1. Precision pipettes
2. Distilled or deionized water
3. EIA kit Microplate Washer
4. EIA kit Microplate Reader with a 450 nm and a 620 - 650 nm filter
Cryptosporidium ELISA kit Background Information:
Cryptosporidium is a coccidian parasite that is recognized as an important enteric pathogen. The organism causes an acute, though self-limiting infection in immunocompetent individuals. Incubation periods of 1 to 12 days have been reported with most oocyst shedding ending by day 21. Symptoms range from mild to severe diarrhea with a variety of complications. The infection in immunocompromised patients is much more severe and may often be life threatening. Passage of fluid, up to 12 liters per day, has been reported. Multiple pathways of Cryptosporidium transmission have been implicated. These include animal to human, water contamination and person-to-person. The latter may include contact between members of the same household, day care centers, and homosexual men. Diagnosis of Cryptosporidium infections was done originally by direct detection techniques. However, this method relies on an experienced technician and subsequent observation of intact organisms. One important alternative has been the development of an antigen capture ELISA for use with stools with comparable sensitivity to experienced microscopic examinations.
Cryptosporidium ELISA Test Principle:
During the first incubation, Cryptosporidium specific antigen present in the stool specimens are captured by antibodies attached to the microwells. The wells are incubated and washed before anti Cryptosporidium antibodies conjugated to horseradish peroxidase are added. The enzyme conjugate will sandwich any antigen bound to the wells. After washings to remove unbound enzyme, a chromogen is added which develops a blue color in the presence of the enzyme complex. The stop solution ends the reaction and turns the blue color to yellow.
Notable features of the Parasitology ELISA Kits:
User-friendly directions and explanation of test procedures
Simple and safe reagent preparation
Clear instructions on specimen collection
Comprehensive package of required materials
Explicit quality control and storage guidelines
Reliable and easy-to-read test results
Product inserts for most Parasitology ELISA kits follow a similar method. See the Cryptosporidium ELISA Kit product insert for details on preparation, procedures, quality control, and test result interpretation.
Diagnostic Automation also provides other Parasitology ELISA Kits. For more information about these ELISA Kits or Rapid Tests, IFA Kits, CLIA Test Kits, or Serology tests, please see our website home page, or contact our Customer Service Representative at 818-591-3030.
Product Note:
Cryptosporidium ELISA Test Kit is for the qualitative determination of Cryptosporidium in feces.
ELISA kits - Rapid tests- Drug tests- Pregnancy test - IFA kits - CLIA assays - Serology tests - Instrumentation
©1992 Diagnostic Automation/Cortez Diagnostics Inc. All rights reserved.