
ELISA Kits
- • Anemia ELISA kits
- • Allergy ELISA kits
- • Autoimmune Disease kits
- • Bone Metabolism ELISA kits
- • Blood bank ELISA kits
- • Cancer ELISA kits
- • Cardiac Markers ELISA kits
- • Diabetes Assays ELISA kits
- • Drug test ELISA kits
- • Fertility ELISA kits
- • Food ELISA kits
- • Infectious Disease ELISA kits
- • Other ELISA Kits
- • Parasitology ELISA kits
- • Steroid ELISA kits
- • Thyroid ELISA kits
Rapid Tests
- • Allergy Rapid tests
- • Bone Metabolism
- • Cancer Rapid tests
- • Cardiac markers Rapid tests
- • Drug Tests
- • Fertility Rapid tests
- • Hepatitis Panel
- • Infectious Disease & other tests
- • Other
- • Ovulation Rapid tests
- • Pregnancy tests
- • Urine Reagent Strips tests
IFA Kits
Chemiluminescence Immuno Assays
- • Allergy Assays
- • Autoimmune Thyroid Assays
- • Cardio-Vascular Monitoring
- • Diabetes Assays
- • Fertility Assays
- • Growth Deficiency
- • Infectious Disease Assays
- • Others
- • Steroid Assays
- • Thyroid Assays
- • Tumor Marker Assays
Serology Tests
- • ASO (Anti-Streptolysin-O)
- • CRP (C-Reactive Protein)
- • Mono (Infectious Mononucleosis)
- • RF (Rheumatoid Factor)
- • RPR (Rapid Plasma Reagin)
- • SLE (Systemic Lupus Erythematosus)
Instrumentation



H. Pylori Antigen Rapid Test (Stool)
| Name |
H. Pylori Antigen Rapid Test Kit |
|---|---|
| Full name |
Human Helicobacter Pylori Rapid Test (Cassette) Antigen RapiCard InstaTest, Export Use Only |
| Category Name | Infectious Disease & other tests |
| Test | Bulk or Boxed in 25 or 50 tests |
| Specificity | 98.4% |
| Sensitivity | 98.8% |
 |
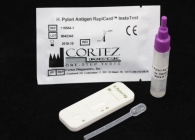 |
 |
H. Pylori Antigen Rapid Test (Stool) description:
"Export Use Only"
The OneStep H. pylori Antigen Rapid Test is an in vitro qualitative immunochromatographic assay for the rapid detection of Helicobacter pylori antigens in human stool specimen. The test results are intended to aid in the diagnosis of H. pylori infection, to monitor the effectiveness of therapeutic treatment and to confirm the eradication of H. pylori in peptic ulcer patients.
Materials Provided with H. Pylori stool Rapid Test kit:
1. OneStep H. pylori Antigen Rapid Test
Each cassette contains a test strip with H. pylori specific antibody on
test region of the membrane and colored H. pylori antibodygold
conjugate pad
2. Sample bottle: contains 1.5 ml of stool specimen collection buffer
3. Instruction for use
Materials required but not provided:
1. Specimen collection container
2. Clock or timer
H. Pylori Rapid Test kit Background Information
Helicobacter pylori is a corkscrew-shaped, gram-negative rod that lives in the mucous layer of the stomach. H. pylori infection is now accepted as the most common cause of gastritis, and is etiologically involved in gastric ulcer, duodenal ulcer, gastric adenocarcinoma and primary gastric B-cell lymphoma. The organism is very common, infected at least half of the world population. H. pylori infection is typically acquired in childhood. Once acquired, infection persists chronically, probably continuing in the stomach throughout life. The damage to gastric structure and function of stomach is constant and direct. Approximately one in six of H. pylori infection develops peptic ulcer disease and a small portion of H. pylori infection leads to gastric cancer. The diagnostic tests for H. pylori can be classified into two categories: Invasive and Noninvasive tests. Direct detection by invasive test procedures requires an endoscopy and biopsy specimens from antrum and stomach body .The presence of H. pylori is then confirmed by direct culture, histological examination or rapid urease test. The endoscopy and biopsy specimens offer direct detection of active H. pylori infections. Although the procedure is highly specific and high positive predictive value, the cost and discomfort to the patients are very high. The most widely available noninvasive test is probably the serological based test. The serology test detects H. pylori specific IgG antibody in patient serum with current or prior infection. Serology test is a simple, convenient test with relative high sensitivity. The main limitation of serology test is the inability to distinguish current and past infections. Antibody may be present in the patient serum long after eradication of the organism. The urease breath test (UBT) with C or C labeled urea, is a noninvasive test based on the urease activity of the organism. UBT detects active H. pylori infection and is highly sensitive and specific. The UBT requires a high density and active bacteria and should not be performed until 4 weeks after therapy to allow residual bacterial to increase to a sufficient number for detection.
H. Pylori Antigen Rapid Test Principle .
OneStep H. pylori Antigen RapiCard InstaTest is a sandwich solid phase immunochromatographic assay. To perform the test, an aliquot of diluted stool sample is added to the sample well of the test cassette. The sample flows through a label pad containing H. pylori antibody coupled to red colored colloidal gold. If the sample contains H. pylori antigens, the antigen will bind to the antibody coated on the colloidal gold particles to form antigen-antibody-gold complexes. These complexes move on the nitrocellulose membrane by capillary action toward the test line region on which H. pylori specific antibodies are immobilized. As the complexes reach the test line, they will bind to the antibody on the membrane in the form of a line. A second red control line will always appear in the result window to indicate that the test has been correctly performed and the test device functions properly. If H. pylori antigen is not present or lower than the detection limit of the test, only the control line will be visible. If the control line dose not developed, the test is invalid. For additional details please refer to the instructions for use.
Notable features of H. pylori Rapid Test:
-Reliable and inexpensive
-Convenient and easy-to-understand directions
-Clear preparation procedures
-Simple and safe specimen collection directions
-Comprehensive package of required materials and components
-Rapid and accurate test results
See H. pylori Stool Rapid Test Product Insert for specific details on preparation, procedures, quality control, and interpretation of test results.
H. Pylori Rapid Test Results Interpretation
The result is positive if both the C line and T line appear, indicating that the IgG antibodies specific to H. pylori are detected. If only the C line appears in the control region, the test indicates that no antibodies to H. pylori are detected and the result is negative. For additional details please refer to the instructions for use.
H. Pylori Rapid Test Kit Performance Characteristics
Sensitivity and Specificity:
This h. pylori test kit was evaluated on 170 adults. The test results were compared to diagnosis of H. pylori infection by reference tests, urease breath test and histology tests. Patients were considered positive if both rapid urease and histology tests were positive. Patients with both negative urease breath test and histology tests were considered negative. Among 50 positive samples and 120 negative samples, OneStep H. pylori Antigen Rapid Test showed 94.0% clinical sensitivity and 96.7% specificity. The accuracy is 97.5%.
Reproducibility:
Reproducibility of OneStep H. pylori Antigen Rapid Test was determined using negative, low positive, and high positive samples along with negative and positive controls. These samples were tested in replicates of 8 in a blind study by 5 operators working independently in the same laboratory. The agreement of the expected result was 100%.
Diagnostic Automation Inc. also offers other Infectious Diseases Rapid tests, and Infectious Diseases ELISA test kits. For more information about Rapid tests, CLIA tests, IFA kits, or ELISA test kits, please contact our Customer Service Representatives at 818-591-3030 .
H. Pylori Related Kits:
H. Pylori Rapid Test Kits
H. Pylori Rapid Test (Serum Cassette) 118561-1
http://www.rapidtest.com/index.php?i=H.-Pylori-Serum-Rapid-Test-&id=387&cat=26
H. Pylori Rapid Test (Cassette Serum / Whole Blood Test) 118562-1
http://www.rapidtest.com/index.php?i=H.-Pylori-Rapid-Test-(Serum-&-Whole-Blood-)-&id=668&cat=26
H. pylori ELISA Kits
H. pylori IgA ELISA kit
http://www.rapidtest.com/index.php?i=H.-pylori-IgA-ELISA-Test&id=141&cat=16
H. pylori IgG ELISA kit
http://www.rapidtest.com/index.php?i=H.-pylori-ELISA-Test&id=140&cat=16
H. pylori IgM ELISA kit
http://www.rapidtest.com/index.php?i=H.-pylori-IgM-ELISA-kit&id=142&cat=16
H. Pylori Antigen ELISA kit
http://www.rapidtest.com/index.php?i=H.-Pylori-Antigen-ELISA-kit&id=613&cat=16
Additional H. pylori kits available are:
H. pylori IgA CLIA, H. pylori IgG CLIA & H. pylori IgM CLIA Kit
H. pylori CLIA Kits
http://www.rapidtest.com/index.php?product=Infectious-Disease-Assays&cat=39
Diagnostic Automation Inc. also provides other Rapid Test Kits:
http://www.rapidtest.com/index.php?product=Rapid-Tests
For more information about Rapid Tests, ELISA Kits, CLIA tests, or IFA Tests, please contact our Customer Service Representatives at 818-591-3030.
*Not to be sold, shipped, or distributed in the United States.
Product Note:
OneStep H. pylori Antigen Rapid Test
is an Immunochromatographic assay that uses antibody- coated colloidal gold to detect
the presence of H. pylori antigens in stool specimens. The test detects
directly antigens in specimens for an active infection. The test is simple and
easy to perform and the test results can be visually interpreted within 10
minutes.
ELISA kits - Rapid tests- Drug tests- Pregnancy test - IFA kits - CLIA assays - Serology tests - Instrumentation
©1992 Diagnostic Automation/Cortez Diagnostics Inc. All rights reserved.


 Strep A Rapid Test
Strep A Rapid Test
 Malaria pf/pv Rapid Test
Malaria pf/pv Rapid Test
 Syphilis Rapid Test
Syphilis Rapid Test
 Leishmania (VL) Rapid Test
Leishmania (VL) Rapid Test
 Salmonella Rapid Test
Salmonella Rapid Test








